The open-shell theory described here is restricted by two simplifying assumptions: (1) the total energy is expressible in terms of the shell-averaged one-electron, coulomb, and exchange integrals already defined [14] for closed shells, and (2) the total energy can be written in terms of only one open-shell density matrix [15].
Closed shells will be indexed by k or ![]() , open shells by m or n, and
i or j will index either type. Shell occupation numbers will
correspondingly be
, open shells by m or n, and
i or j will index either type. Shell occupation numbers will
correspondingly be ![]() ,
, ![]() , etc.
, etc. ![]() is
always
is
always ![]() . The closed-shell, open-shell, and total density matrices
are
. The closed-shell, open-shell, and total density matrices
are
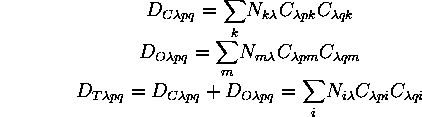
The integrals over MOs are
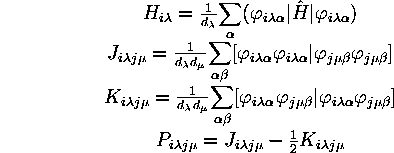
The one-electron energy for both closed and open shells will be given simply by the product of the occupation numbers and the one-electron MO integrals. The electron repulsion energy between closed shells will be of the same form as in a closed-shell state:
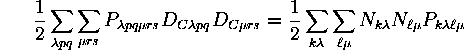
The electron repulsion energy between a closed shell and an open shell also depends only on occupation numbers and P integrals. Since the closed shell is totally symmetric, it does not matter in what way the open-shell orbitals are occupied. Only the number of open-shell electrons is needed. This corresponds to the fact that only one summation is needed in the shell averaging of coulomb and exchange integrals. The resulting value is independent of which value of the unaveraged index is used. The (closed-shell)-(open-shell) interaction energy is thus
![]()
The electron repulsion energy between open shells depends on the spin and spatial coupling of the occupied spinorbitals in the wavefunctions for the various terms. This energy cannot always be written in terms of shell-averaged coulomb and exchange integrals, but since these are the only integrals being considered in this treatment, only those cases where the open-shell interaction energy can be written in terms of these two types of integrals are considered here:
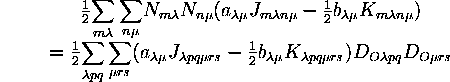
The ![]() and
and ![]() coefficients are constrained not to
depend on the shell indices m and n so that the energy can be expressed in
terms of a single open-shell density matrix, as desired.
coefficients are constrained not to
depend on the shell indices m and n so that the energy can be expressed in
terms of a single open-shell density matrix, as desired.
Thus, within the restrictions stated, the total energy is
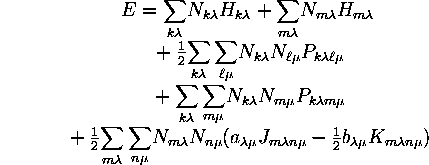
Since the (closed-shell)-(closed-shell) and (closed-shell)-(open-shell) terms are of the same form, both of them are contained in a single sum over all shells. Such a sum also includes an (open-shell)-(open-shell) part which must be subtracted out.
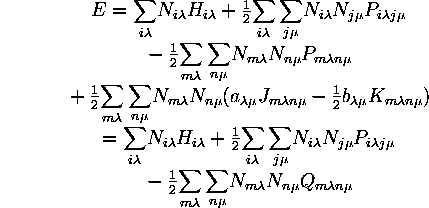
where
![]()
and
![]()
( ![]() and
and ![]() should not be confused with the
use of
should not be confused with the
use of ![]() and
and ![]() as symmetry subspecies indices.)
as symmetry subspecies indices.)
In terms of basis functions the total energy expression is
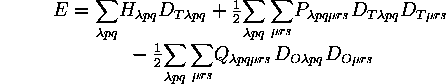
where
![]()