|
|
Photoreceptor Dynamics
| Home | ||||
Research
|
||||
| People | ||||
| Publications | ||||
| Photo Gallery | ||||
| Collaborations | ||||
| Positions | ||||
| Courses | ||||
| Contact | ||||
| Group Internal | ||||
|
Last Updated: 27 Oct 2008 |
|
We are currently investigating a recently discovered blue-light photoreceptor cryptochrome, a sequence analogue of photolyase, but having a totally different function. This flavoprotein uses photon energy to synchronize the circadian clock. We are characterizing initial signaling-state formation and transduction in these complexes with state-of-the-art femtosecond spectroscopy. Dynamics and function are well synchronized by femtosecond pulse initiation and their temporal evolution will be directly probed. Complex recognition and conformational changes will be studied at longer time scales. Abnormal clock function causes a variety of diseases and syndromes and these studies will reveal molecular mechanisms of biological timing. |
|
Ultrafast dynamics and anionic active states of the flavin cofactor in cryptochrome and photolyase.
Y.-T. Kao, C. Tan, S.-H. Song, N. Ozturk, J. Li, L. Wang, A. Sancar and D. Zhong, J. Am. Chem. Soc. 130, 7695 (2008).
[Web link]
[PDF]
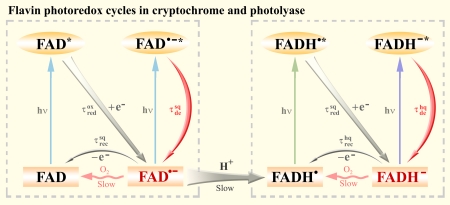
Ultrafast dynamics of flavins in five redox states.
Y.-T. Kao, C. Saxena, T.-F. He, L. Guo, L. Wang, A. Sancar and D. Zhong, J. Am. Chem. Soc. 130, 13132 (2008).
[Web link]
[PDF]

|
